


This study demonstrates a single-exposure quantitative phase imaging approach that enables millisecond-scale observation of intracellular dry mass dynamics in live cells. By integrating a cascaded dual non-polarizing beam splitter architecture with a high-performance sCMOS camera (Revealer Gloria 4.2), four phase-shifted interferograms are acquired simultaneously within one frame. The experimental results establish that the sCMOS camera functions not only as an imaging sensor but as a temporal synchronization core, enabling high-throughput, low-noise, and quantitatively reliable phase reconstruction in dynamic biological systems.
Quantitative phase imaging (QPI) provides label-free measurement of optical thickness, refractive index, and dry mass distribution in biological samples, making it a fundamental tool for studying live-cell dynamics. However, conventional spatial light interference microscopy relies on sequential acquisition of multiple phase-shifted frames, which fundamentally limits temporal resolution and prevents accurate capture of fast subcellular events occurring at the millisecond scale.
Existing single-exposure solutions, including polarization multiplexing and diffraction-based splitting, improve acquisition speed but often compromise space-bandwidth product or introduce intensity imbalance among channels, thereby reducing signal-to-noise ratio and measurement fidelity. These limitations directly impact the reliability of quantitative phase reconstruction in dynamic systems.
To overcome these constraints, the present study develops a single-exposure spatial light interference microscopy (SE-SLIM) system based on cascaded non-polarizing beam splitters. This configuration ensures equal intensity distribution across four optical paths while preserving spatial resolution. Crucially, the introduction of a high-frame-rate, low-noise sCMOS camera enables simultaneous acquisition of multi-channel interferometric information within a single exposure, eliminating temporal mismatch errors that are inherent to sequential imaging systems.
The imaging system integrates a commercial bright-field microscope with a quantitative phase module, in which the sCMOS camera serves as the central data acquisition unit. The Revealer Gloria 4.2 sCMOS camera provides a resolution of 2048 × 2048 pixels with a pixel size of 6.5 μm and operates at 135 frames per second under full-frame conditions. Its back-illuminated scientific CMOS architecture delivers a quantum efficiency of up to 95% and a read noise as low as 1.2 electrons, enabling precise detection of weak interferometric fringes.
The optical configuration is built upon a broadband halogen illumination source coupled with an annular illumination module. The incident light is first divided into two beams using a non-polarizing beam splitter and a right-angle prism. A second identical stage further splits each beam into two, resulting in four parallel beams with equal intensity. A spatial light modulator applies discrete phase shifts of 0°, 90°, 180°, and 270° to these beams. After propagation through a Fourier lens, four spatially separated interferograms are formed on the sensor plane of the sCMOS camera.
Within this architecture, the sCMOS camera is not merely a detector but the only component capable of synchronously capturing all phase-shifted signals in a single exposure, thereby enabling true parallel phase acquisition.
The imaging strategy combines parallel four-step phase shifting with single-frame acquisition. The spatial light modulator introduces controlled phase modulation across the four optical channels, while the sCMOS camera records all corresponding intensity distributions simultaneously. This ensures that all phase information is sampled under identical temporal conditions.
The captured images are processed using a SURF-based segmentation and registration algorithm implemented in MATLAB. Phase reconstruction is subsequently performed using an arctangent-based calculation. Because all input data originate from a single exposure, the reconstruction process is inherently free from inter-frame phase drift, which is a common limitation in conventional systems.
Dynamic imaging experiments are conducted on red blood cells and HeLa cells under DMSO stimulation. The sCMOS camera records high-speed temporal sequences, providing a continuous dataset that directly supports quantitative analysis of morphological evolution and intracellular dry mass redistribution.
Revealer Gloria 4.2 sCMOS camera provides direct observational evidence for system calibration, phase reconstruction and dynamic live-cell imaging.
4.1 Temporal Synchronization and Single-Frame Acquisition
In conventional SLIM systems, four independent exposures are required to obtain a complete set of phase-shifted data. In contrast, the sCMOS camera employed in this study captures all four channels within a single frame at 135 fps, effectively increasing temporal resolution by a factor of four. This single-frame acquisition eliminates phase errors caused by sample motion or illumination fluctuations between frames.
The experimental observations demonstrate that the sCMOS camera acts as a temporal unifier, ensuring that all phase information is acquired under identical conditions. This capability is essential for accurate phase reconstruction in dynamic biological systems.
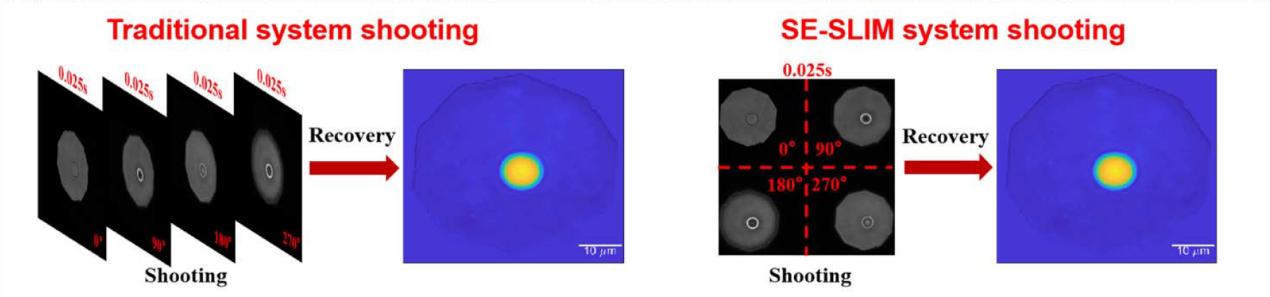
Figure 1 Single-exposure four-channel synchronous acquisition mechanism of sCMOS camera. Compared with the traditional multi-frame acquisition process, it demonstrates unified temporal sampling capability.
4.2 Phase Sensitivity and System Stability
System calibration results indicate that the phase resolution is better than 500 nm, with spatial phase sensitivity reaching 1.84 nm and temporal phase sensitivity reaching 0.82 nm. Continuous acquisition at 256 fps shows that the temporal distribution of phase values remains highly stable, with no observable drift or abrupt noise fluctuations.
These results confirm that the low read noise and high quantum efficiency of the sCMOS camera directly translate into nanometer-scale phase sensitivity. The camera’s stability enables long-duration, high-speed acquisition without degradation of measurement accuracy.
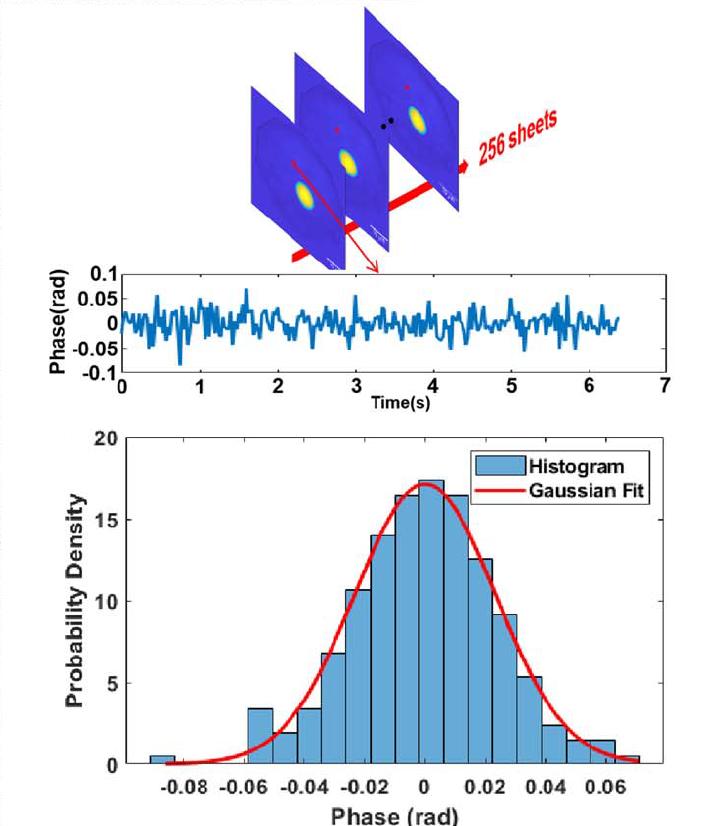
Figure 2 Statistical results of phase noise based on high-speed continuous acquisition by sCMOS camera, illustrating temporal phase stability and the origin of nanoscale sensitivity.
4.3 Quantitative Accuracy Validation Using Standard Samples
Quantitative imaging of 10 μm polystyrene microspheres demonstrates the accuracy of the system. The reconstructed average diameter is 10.48 μm, with an error below 4%, while the measured phase peak deviates by less than 4% from theoretical predictions.
The sCMOS camera captures interferometric signals with high fidelity, without signal loss or distortion, providing a reliable basis for quantitative phase reconstruction even under weak signal conditions.

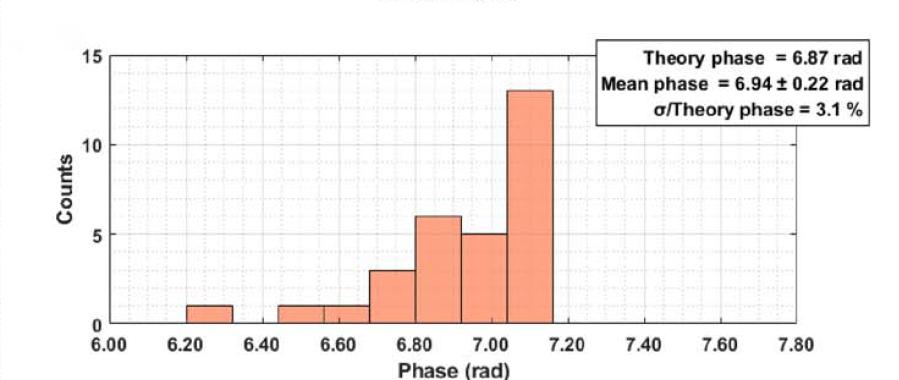
Figure 3 Statistical results of microsphere interference signals and phase reconstruction consistency acquired by the sCMOS camera, verifying its linear response and stability in quantitative measurement.
4.4 Dynamic Imaging of Live Cells
High-speed imaging at 135 fps over a continuous duration of 6.4 seconds enables detailed observation of dynamic cellular processes. The system captures osmotic swelling and morphological flattening of red blood cells, as well as contraction, membrane rupture, and vesicle formation in HeLa cells under DMSO stimulation.
These results demonstrate that the sCMOS camera enables real-time, label-free, and quantitative monitoring of cellular dynamics. Its ability to provide high-frame-rate temporal data is critical for resolving transient biological events that occur on the millisecond timescale.
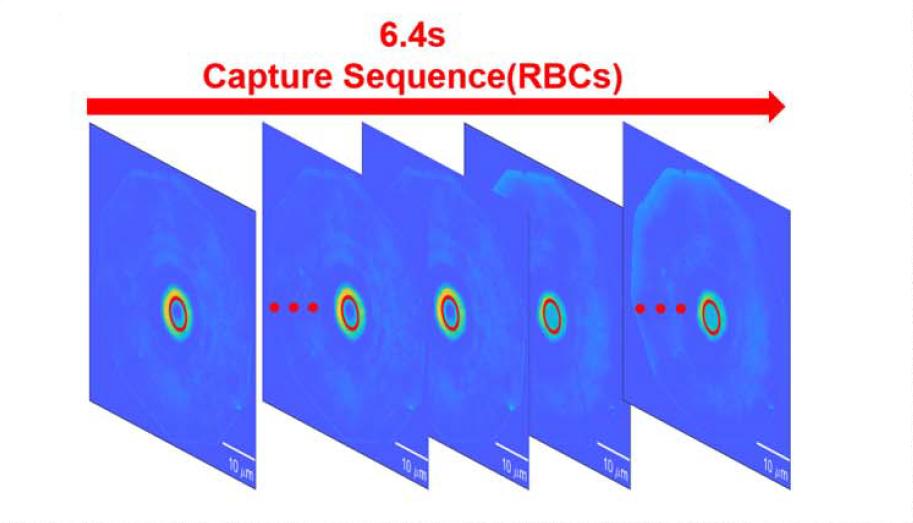
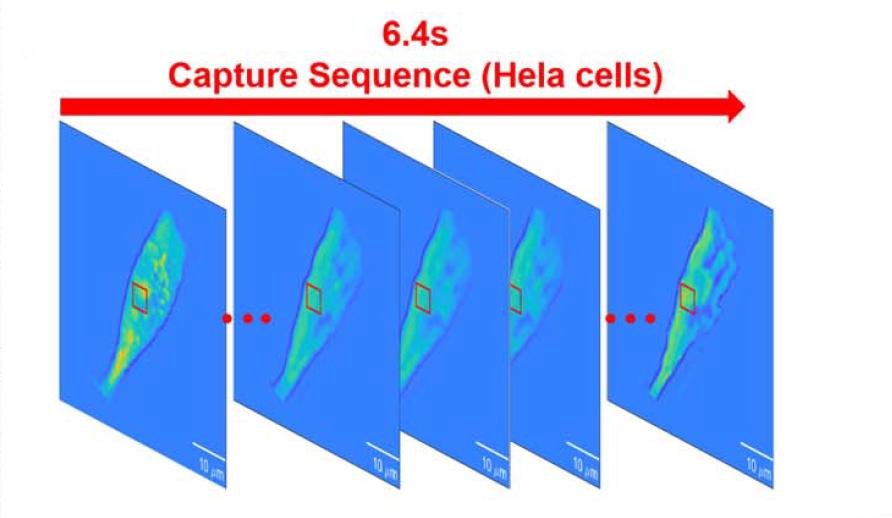
Figure 4 Dynamic cell sequences and dry mass evolution curves continuously acquired by the sCMOS camera at 135 fps, reflecting temporal continuity in observing high-speed biological processes.
This study establishes a single-exposure SE-SLIM system in which the sCMOS camera plays a central and irreplaceable role in determining system performance. By enabling simultaneous acquisition of four phase-shifted interferograms within one frame, the camera fundamentally improves temporal resolution and eliminates inter-frame phase errors.
The high frame rate allows complete capture of millisecond-scale cellular dynamics, while the low noise and high dynamic range ensure reliable detection of weak interferometric signals. The large sensor format preserves sufficient spatial sampling even after segmentation into multiple channels, supporting accurate subpixel registration and quantitative reconstruction.
Taken together, the results demonstrate that the sCMOS camera is not merely an imaging component but the key enabling technology that defines throughput, sensitivity, and dynamic measurement capability in high-speed quantitative phase imaging systems. This work provides a reproducible, device-level solution for real-time, label-free observation of live-cell dynamics.
 English
English Deutsch
Deutsch






